

Despite efforts across the globe to move toward a circular plastics economy, more than three quarters of the 400 metric tons of plastic produced worldwide each year still ends up as waste.
A group of chemistry researchers at the University of Illinois Urbana-Champaign demonstrated a way to use the renewable energy source of electricity to recycle a form of plastic that’s growing in use but more challenging to recycle than other popular forms of plastic.
In their study recently published in Nature Communications, they share their innovative process that shows the potential for harnessing renewable energy sources in the shift toward a circular plastics economy.
“We wanted to demonstrate this concept of bringing together renewable energy and a circular plastic economy,” said Yuting Zhou, a postdoctoral associate and co-author, who worked on this groundbreaking research with two professors in chemistry at Illinois, polymer expert Jeffrey Moore and electrochemistry expert Joaquín Rodríguez-López.
The project was conceived by Moore who had experience working with Poly(phthalaldehyde), a form of polyacetal. Polyoxymethylene (POM) is a high-performance acetal resin that is used in a variety of industries, including automobiles and electronics. A thermoplastic, it can be shaped and molded when heated and hardens upon cooling with a high degree of strength and rigidity, making it an attractive lighter alternative to metal in some applications, like mechanical gears in automobiles. It is produced by different chemical firms with slightly different formulas and names, including Delrin by DuPont.
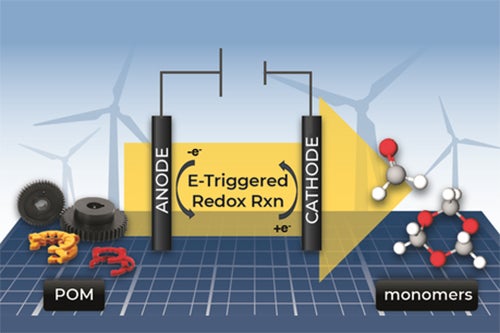
When recycling, those the highly crystalline properties of POM make it difficult to break down. It can be melted and molded again, but POM’s original material properties are lost, limiting the usefulness of the recycled material.
“When the polymer was in use as a product, it’s not a pure polymer. It will also have other chemicals like coloring additives and antioxidants. So, if you simply melt it and remold it, the material properties are always lost,” Zhou explained.
The Illinois research team’s method uses electricity, which can be drawn from renewable sources, and takes place at room temperature.
This electro-mediated process deconstructs the polymer, breaking it down into monomers — the molecules that are bonded to other identical molecules to form polymers.
Significant chemical repurposing breakthroughs have been achieved in the past five years on commonly known synthetic plastics, like PE, PET, and PS.
However, there has been limited experimentation with the more challenging POM, particularly with electricity as the driver of the process.
In the study, the researchers demonstrate their methods using Delrin. They begin by dissolving small beads of the polymer in a liquid to loosen the bonds in the polymer chain. In the next phase of electrocatalysis, the Delrin polymer chains are broken down into monomer form.
The key, Zhou said, is the organic solvent that dissolves the plastic. She tested many organic solvents, starting with rubbing alcohol, but discovered only one organic that worked, Hexafluoroisopropanol, commonly abbreviated HFIP.
“It’s not like immediately. It had to sit for several hours,” said Zhou, who explained that they discovered that not only does the HFIP dissolve the polymer, but it likely plays a key role in the depolymerization in the electrocatalysis phase. “It’s also functions as a proton donor (catalyst) with applied potential.”
Basically, the HFIP generates acid during the electrolysis, and the acid, the researchers believe, is what is breaking down the polymer to monomers.
“That’s what we hypothesize is happening,” she said. “There may be other potential depolymerization mechanisms. But with all the experimental evidence that we collected, it’s more leaning toward an electro-mediated acid depolymerization process.”
After successfully demonstrating their process on small beads of pure POM, they wanted to try a commercial product made of Delrin to further test their method, so they used keck clip sheds, which are commonly used in chemistry labs and made of Delrin. And again, the process worked.
“At least personally to me, this work is more like an encouragement to me that you’re not thinking crazy to use electricity to break down plastic. It’s possible. It’s very challenging. There are limitations. There are a lot of walls that we will hit. But it’s possible,” Zhou said. “We can use electricity to break down a real manufactured product.”
Zhou said they want to further explore the power of electrocatalysis in plastic deconstruction by experimenting on more challenging types of plastic, targeting the selective upcycling of POM to formic acid and adopting this method into a flow system.
“We also are hoping that this work can inspire other engineers and synthetic chemists, and the field to probe and think about using electricity to deconstruct synthetic plastic,” Zhou said.
This research was financially supported by the Joint Center for Energy Storage Research, an Energy Innovation Hub funded by the U.S. Department of Energy.
Moore is Stanley O. Ikenberry Research Professor, professor emeritus of chemistry, and Howard Hughes Medical Institute Professor and affiliated with Center for Advanced Study, Beckman Institute, Materials Science and Engineering, Materials Research Lab, Carle Illinois College of Medicine, and Carl R. Woese Institute for Genomic Biology. Rodríguez-López is J. Andrew and Susan S. Langan Professorial Scholar, LAS Distinguished Professorial Scholar, and professor of chemistry. He is also associated with Beckman and the Materials Research Lab.


